This post continues from our last post on this topic: Glassware Cleaning Part 2 — How do you know it’s clean? This post can be found here.
In our last post, we reviewed the basic concept of cleaning formulation equipment and presented a basic process that could be used for clean-up. This thought process generally works well with simple, ideal formulations that are readily cleaned. However, as every beginning scientist knows, formulations can get complicated quickly. So, what about when it gets more complicated?
An excellent book chapter for the beginning scientist is “Chapter 5: Validation of the Cleaning Process” found in “Handbook of Pharmaceutical Manufacturing Formulations : Sterile Products by S. K. Niazi”, and this source has greatly informed this post. First and foremost, this chapter emphasizes a concept that we also emphasize here in this blog — start at the end! The end-point here is determining when something is clean, so we need to determine what that is first before going into great detail on the cleaning process and analytics. In fact, this chapter mentions that many mistakes in the industry are made on this point, and that many cleaning programs involve extensive procedures and analytics without ever stating a real definition of what clean is for the equipment involved!
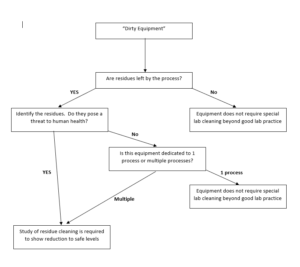
To avoid such mistakes, let’s define what “clean” will be for our work. The thinking here is that we need to look at the purpose for the equipment, the potential for residues to affect future batches, and whether the equipment is shared or dedicated between processes. A good way to think through this is a decision tree for your equipment. An example is shown below:
Again, the point of working through this thought process is to determine what the end goal is. Anything ending in a goal of cleaning to good lab practice, simply means cleaning to a level descried by your typical SOPs at your place of work. Many times this is simply clean until visibly clean, 3x rinse with water, etc. Alternatively, any time that they are significant residue levels left that pose a threat to human health or process integrity, we must perform studies to determine the level of clean that we need. Admittedly, this example decision tree is simplistic, and you will need to develop it significantly for your needs. However, it illustrates the basic concept that we must have a decision on the type and level of “clean” needed within a broader framework and it must be well-thought out. Remember, we have to start at the end!
Once your final goal is established, we can start to work backwards through the cleaning process. For the sake of discussion, let’s say that we find we have a multi-process piece of equipment, contaminated with hazardous residues after a process. Our bench-top data suggest that cleaning must take place immediately after process completion, as dried residues will be harder to remove. Let’s further suppose that while rinsing was somewhat effective, it was a case of diminishing returns and that scrubbing until visibly clean will also be a necessary step. What a mess!
We will need to extensively think through this process and this will likely involve working with engineers, quality control, quality assurance, and your company’s safety team. You will need to work backwards as shown below with this thought process:
- Establish the necessary level of clean. This has already been done above.
- Establish how we will know that we are at that level of clean. This will likely involve validation of our analytical method and establishment of sampling plan for the equipment. Keep this plan practical and well-documented.
- Determine the last step of the process. Since scrubbing is required in this example, let’s say that manual cleaning with some type of disposable scrubbing instrument is the last step.
- Know think about how we will get to that last step. Since this is hazardous material, we do not want personnel immediately scrubbing the equipment after the process. However, could we rinse the equipment a certain number of times first to the reduce the hazard? Is there an inactivating agent that we could rinse with? Are there other types of rinse equipment that could be used to enhance rinsing effectiveness (spray nozzles, etc.)?
- And etc. all the way to the beginning of the process.
And how do we test our process at scale? Chapter 5 from S. K. Niazi suggests the use of “Placebo Product” on equipment for at scale testing. Placebo product is defined as a formulation w/ out the active ingredient (or w/ an easily traceable but non-hazardous active ingredient such as naproxen sodium or a fluorescent tracer) put through the process equipment that will be used for batch manufacturing in the future. Doing this allows the formulation scientists to check a number of things including: how residues are distributed on the equipment, how effective the cleaning process is at various spots on the equipment, and the actual analytical power of the analytical method when samples are gathered in situ rather than in an ideal lab sampling. Depending on the exact situation, it may also be worthwhile to perform an engineering run with actual product, as well, but again this is highly situationally dependent.
And lastly, two more bits of wisdom from S. K. Niazi in Chapter 5. First, be sure that any cleaning agents used are also easily cleanable! This may seem obvious, but in many cases this can be totally forgotten. Leaving such agents on the equipment can be just as problematic as leaving formulation on the equipment. Secondly, while tempting, DO NOT TEST UNTIL CLEAN! It may be tempting to have a procedure saying: rinse 2 – 5 times with sampling after each time and stop when it is clean. However, the problem here is that this procedure is not well thought out scientifically. Why would sometimes 2 rinses clean the equipment but other times 5 rinses clean the equipment? All procedures should be based on data, not guesswork, and oddities should be thoroughly investigated.
For the next entry in this series, I’d like to tackle some realistic problems! Go ahead and suggest a problem you are working on or thinking about in the comments! Look forward to seeing what you guys have!
References:
[1] Chapter 5: Validation of the Cleaning Process from Handbook of Pharmaceutical Manufacturing Formulations : Sterile Products by S. K. Niazi